ARTICULO DE REVISIÓN
REVISTA DE LA FACULTAD DE MEDICINA HUMANA 2021 - Universidad Ricardo Palma10.25176/RFMH.v22i1.4129
EXERCISE AS A PROTAGONIST IN MUSCLE PLASTICITY AND IN THE MUSCLE AS AN ENDOCRINE ORGAN: IMPLICATIONS IN CHRONIC DISEASES
EL EJERCICIO COMO PROTAGONISTA EN LA PLASTICIDAD MUSCULAR Y EN EL MÚSCULO COMO UN ÓRGANO ENDOCRINO: IMPLICACIONES EN LAS ENFERMEDADES CRÓNICAS
Amelia Vargas-Pacheco 1, a, Lucy E. Correa-López 2, b
1 Universidad Nacional Federico Villarreal. Lima, Perú
2 Facultad de Medicina Humana, Universidad Ricardo Palma. Lima, Perú
a Specialist in health services administration
b Masters in Economy
ABSTRACT
The active skeletal muscle and the maintenance of muscle mass are essential for good health,
wellness and disease prevention and treatment. It has recently been documented that the muscle, as an
endocrine organ, is capable of synthesizing myokines. These peptides are secreted in response to
muscular contractions induced by exercise, and can develop molecular communication with other tissues,
improving cardiovascular, metabolic, and mental health.
The endocrine function of a muscle continues to be in a constant investigation; however,
knowledge up to date urges us to continue promoting physical activity. Additionally, the extended
knowledge of myokines is useful to prescribe individualized physical exercise programs to each patient.
Keywords: Adaptive plasticity, muscles, cytokines, physical exercise, motor activity, chronic disease (Source : MeSH - NLM).
RESUMEN
El músculo esquelético activo y el mantenimiento de la masa muscular son esenciales para la salud, el
bienestar, la prevención y el tratamiento de enfermedades. Recientemente se ha documentado que el
músculo, como órgano endocrino, es capaz de sintetizar mioquinas. Estos péptidos son secretados en
respuesta a las contracciones musculares inducidas por el ejercicio, y son capaces de crear comunicación
molecular con otros tejidos, beneficiando así la salud cardiovascular, metabólica y mental.
La función endocrina del músculo continúa en investigación constante, sin embargo, lo conocido
hasta ahora nos insta a seguir promoviendo la actividad física. Adicionalmente a esto, extender el
conocimiento sobre las mioquinas, es útil para poder prescribir el ejercicio físico de manera
individualizada y a la medida de cada paciente.
Palabras Clave: Plasticidad adaptativa, músculos, citocinas, ejercicio físico, actividad motora, enfermedad crónica (Fuente: DeCS BIREME).
INTRODUCTION
The muscle is a Dynamic tissue implied in movement, posture, respiration (1),
and corporal thermoregulation (2). It is considered the most abundant organ in
the human body (2), since it represents 30 to 50% of total corporal weight
(3).
Since 450 B.C., we have evidenced the positive effects of physical activity in health and the
prevention of chronic diseases. As was cited by “the father of medicine, Hippocrates of Kos: “Walking is
man’s best medicine” and “sports is a preserver of health”. (4) in this sense,
exercise could also be prescribed as treatment for lifestyle related diseases: cardiovascular diseases,
type 2 diabetes, neurodegenerative diseases and cancer (5).
The benefits of exercise for health and wellness of humans have been attributed to innumerable
physiological mechanisms, however, the transcendental role that the muscle possesses in these processes
has been documented in recent years (6).
In recent years, the skeletal muscle has been identified as a endocrine organ, since it is
capable of synthesizing cytokines called myokines, which possess autocrine, paracrine and endocrine
function (7,8). Thanks to the exercise stimulus, these protein
molecules are released to the blood stream, allowing the communication of the muscle not just with
itself but also with other organs such as the brain, bone, pancreas, intestine, liver, adipose tissue,
cardiovascular system, and skin (5), therefore creating a physiological and
unique connection between exercise and metabolism regulation (2).
Taking into account the progressive deterioration of muscle physiology with age, the active
skeletal muscle and the maintenance of muscle mass are essential for the health care, prevention and
treatment of diseases (9). For this reason, this article seeks to carry out a
current literature review about the importance of physical exercise in the preservation of muscle mass
during a lifetime, for prevention as well as for treatment of non-transmissible chronic diseases.
METHODOLOGY
A review of scientific articles in Spanish and English related to the research topic was carried out, which were collected from PubMed, Elselvier and SciELO. On the other hand, we took into account certain criteria for the adequate article selections: that the article title be chosen with the research topic and that it has a maximum of 6 years old, meaning dated from 2015 onward. We excluded the letter to the editors related with the topic.
RESULTS
Out of the 37 articles found, 35 were chosen. One was excluded because it was published before 2015 and the other because it was a letter to the editor. Tables 1 and 2 detail the most relevant results and conclusions of the chosen articles according to the objective of the present study.
Table 1. Selected articles pertaining to muscle and its plasticity
| AUTHORS | TITLE | YEAR | RESULTS/CONCLUSIONS | TYPE OF ARTICLE |
|---|---|---|---|---|
| Josep M. Argilés PhD, Nefertiti Campos PhD, José M. Lopez-Pedrosa PhD, Ricardo Rueda MD, PhD, Leocadio Rodriguez-Mañas PhD | Skeletal Muscle Regulates Metabolism via Interorgan Crosstalk: Roles in Health and Disease | 2016 | The muscle is notably plastic. Physical activity, especially resistance exercise stimulate the protein synthesis and it is the essence for treatment of muscular atrophy. | Review article |
| M. S. Brook, D. J. Wilkinson, B. E. Phillips, J. Perez-Schindler, A. Philp, K. Smith1 and P. J. Atherton | Skeletal muscle homeostasis and plasticity in youth and ageing: impact of nutrition and exercise | 2015 | Exercise continues to be the most efficient therapeutic strategy for treatment of sarcopenia. Furthermore, it increases muscle strength and basal metabolic rate, optimizes blood pressure, lipid profile and insulin sensitivity. | Review article |
| Rosanna Piccirillo | Exercise-Induced Myokines With Therapeutic Potential for Muscle Wasting | 2019 | Physical activity is one of the pathways in which the muscle protects itself from diseases secondary to muscle wasting. | Review article |
| Christoph Hoffmann, Cora Weigert | Skeletal Muscle as an Endocrine Organ: The Role of Myokines in Exercise Adaptations | 2017 | Myokines are crucial for muscle adaptation for the increase in physical condition. | Review article |
| Pablo Jorge, Marcos-Pardo, Noelia González-Gálvez, Abraham López-Vivancos, Alejandro Espeso-García, et all. | Sarcopenia, Diet, Physical Activity and Obesity in European Middle-Aged and Older Adults: The LifeAge Study | 2020 | Low muscle mass and low muscle strength are criteria for determining sarcopenia European guides. Muscle strength is a predictor of mortality and disability in older adults. | Research article cross-sectional study |
| Jonathan C. Mcleod, Tanner Stokes, Stuart M. Phillips | Resistance Exercise Training as a Primary Countermeasure to Age-Related Chronic Disease | 2019 | Additional to cardiorespiratory condition, muscle mass and strength are also associated with the risk of developing cardiovascular disease and greater mortality rate. The evidence presented emphasizes that resistance training is equivalent and even superior to aerobic exercise as the first line of prevention of chronic diseases. | Review article |
| Sandra Maria Barbalho, Uri Adrian Prync Flato, Ricardo José Tofano, Ricardo de Alvares Goulart , Elen Landgraf Guiguer, Cláudia Rucco P. et al. | Physical Exercise and Myokines: Relationships with Sarcopenia and Cardiovascular Complications | 2020 | Physical inactivity and muscle mass reduction lead to an increase in visceral fat – culminating in an imbalance between anti-inflammatory and proinflammatory state- increasing the vicious cycle between sarcopenia, accumulation of adipose tissue and cardiovascular complications. | Review article |
| José Andrés Rubio del Peral, M. Sonia Gracia Josa. | Ejercicios de resistencia en el tratamiento y prevención de la sarcopenia en ancianos. Revisión sistemática | 2017 | Performing resistance exercise is one of the most adequate patterns to prevent and treat sarcopenia, since this type of exercise alone or combined with aerobic training, reach benefits on mass, muscle strength and basic daily activity functions | Review article Systemic review |
| Youngju Choi, Jinkyung Cho, Mi-Hyun No, Jun-Won Heo, Eun-Jeong Cho, Eunwook Chang, Dong-Ho Park, Ju-Hee Kang, Hyo-Bum Kwak | Re-Setting the Circadian Clock Using Exercise against Sarcopenia | 2020 | Exercise could reduce and even reverse loss of mass and muscle strength. | Review article |
| Sanna Vikberg, Niklas Sörlén, Lisa Brandén, Jonas Johansson PhD, Anna Nordström MD, PhD, Andreas Hult PhD, Peter Nordström MD, PhD | Effects of Resistance Training on Functional Strength and Muscle Mass in 70-Year-Old Individuals With Pre-sarcopenia: A Randomized Controlled Trial | 2019 | We demonstrate the importance of progressive increment of training until it reaches high intensity exercise. This significantly improves prevention of falls, fractures, and optimizes general health. | Research article |
| Mª Victoria Palop Montoro, Juan Antonio Párraga Montilla, Emilio Lozano Aguilera, Milagros Arteaga Checa | Intervención en la sarcopenia con entrenamiento de resistencia progresiva y suplementos nutricionales proteicos | 2015 | A total of 147 studies were found which carried out resistance exercise through 45–60-minute sessions, 2-3 times per week, and 3-4 series of 8 repetitions, with increasing intensity. This exercise produced an increase in muscle mass and strength, as well as increase in protein synthesis and muscle fiber size. | Review article |
| Kelly A. Bowden Davies , Samuel Pickles, Victoria S. Sprung, Graham J. Kemp, Uazman Alam, Daniel R. Moore, Abd A. Tahrani and Daniel J. Cuthbertson | Reduced physical activity in young and older adults: metabolic and musculoskeletal implications | 2019 | Physical activity is vital in musculoskeletal health, bone mass maintenance, as well as reducing the physical and metabolic consequences of aging. | Review article |
| Anna Vainshtein, Marco Sandri | Signaling Pathways That Control Muscle Mass | 2020 | The ability to alter the composition of muscle fibers through lifestyle changes exemplify the incredible property of muscle plasticity. | Review article |
| Kedryn K. Baskin, Benjamin R. Winders, Eric N. Olson | Muscle as a ‘‘Mediator’’ of Systemic Metabolism | 2015 | The type of muscle fiber that prevails in the muscle profoundly impacts in the consumption of systemic energy. | Review article |
| Leandro dos Santos, Edilson S. Cyrino, Melissa Antunes, Diana A. Santos, Luís B. Sardinha | Sarcopenia and physical independence in older adults: the independent and synergic role of muscle mass and muscle function | 2017 | Demonstrated that older adults with loss of mass and muscle strength (sarcopenia) had greater risk of losing physical independence after 90 years of age. | Research article cross-sectional study |
| Source: Own elaboration. |
Table 2. Selected articles pertaining to muscle as an endocrine organ
| AUTHORS | TITLE | YEAR | RESULTS/CONCLUSIONS | TYPE OF ARTICLE |
|---|---|---|---|---|
| Mai Charlotte Krogh Severinsen, Bente Klarlund Pedersen | Muscle–Organ Crosstalk: The Emerging Roles of Myokines | 2020 | Myokines are useful biomarkers for monitoring the type and quantity of exercise that is required to prescribe people with chronic diseases, such as cancer, diabetes, or neurodegenerative diseases. | Review article |
| Mark A. Febbraio | Health Benefits of exercise- more than meets the eye! | 2017 | The communication between tissues during physical exercise prevent metabolic diseases, cancer, retinal degeneration, and memory loss. Cathepsin B, osteocalcin and BDNF are highlighted as messengers. | Review article |
| Luana G. Leal, Magno A. Lopes, Miguel L. Batista Jr. | Physical Exercise-Induced Myokines and Muscle-Adipose Tissue Crosstalk: A Review of Current Knowledge and the Implications for Health and Metabolic Diseases | 2018 | Physical activity is known for optimizing metabolic health through the adaptation of diverse tissues, such as muscle and adipose. | |
| Brian P. Carson | The Potential Role of Contraction-Induced Myokines in the Regulation of Metabolic Function for the Prevention and Treatment of Type 2 Diabetes | 2017 | Resistance exercise releases important concentrations of IL-6, IL-15, BDNF and irisin, which confirms the inclusion of this type of exercise for the protocols of prevention and treatment of DM2. | Review article |
| Julien Delezie, Christoph Handschin | Endocrine Crosstalk Between Skeletal Muscle and the Brain | 2018 | BDNF and PGC-1α play a protagonist role in the synaptic transmission, neurogenesis, memory and in moods. | Review article |
| Jenny Hyosun Kwon , Kyoung Min Moon,* and Kyueng-Whan Min | Exercise-Induced Myokines can Explain the Importance of Physical Activity in the Elderly: An Overview | 2020 | Aerobic and anaerobic exercise generate benefits in the muscle through the release of myokines, which are divided depending on the physical activity. Aerobic: apelin, BAIBA, IL-15, IL-6, irisin, SDF-1, sestina, SPARC, and VEGF-A. Anaerobic: BMP-7, decorin, IGF-1, IL-15, IL-6, irisin y VEGF-A. | Review article |
| Chen He, Wenzhen He, Jing Hou, Kaixuan Chen, Mei Huang, Mi Yang, Xianghang Luo, Changjun Li | Bone and Muscle Crosstalk in Aging | 2020 | Muscle contraction secretes BDNF to improve learning capacity and memory, irisin released by exercise stimulation promotes thermogenesis and increases bone mass. | Review article |
| Christoph Hoffmann, Cora Weigert | Skeletal Muscle as an Endocrine Organ: The Role of Myokines in Exercise Adaptations | 2017 | Myokines play a central role regulating the interaction of myofibrils, immune cells, fibroblasts, and endothelial cells. | Review article |
| Svenia Schnyder, Christoph Handschin | Skeletal muscle as an endocrine organ: PGC-1α, myokines and exercise | 2015 | IL-6 expression is correlated with muscle contraction, promotes gluconeogenesis and lipolysis of adipose tissue. Furthermore, it contributes to the adequate use of energy substrates promoting muscle contraction and affecting distal organs. IL-6 has been named the “exercise factor” (contraction factor) | Review article |
| Marie Lund Bay, Bente Klarlund Pedersen | Muscle-Organ Crosstalk: Focus on Immunometabolism | 2020 | Interventional studies have demonstrated that a reduced number of daily steps generate visceral adipose tissue accumulation, on the contrary physical exercise are associated with a decrease in visceral fat, IL-6, irisin and "meterolin-like" intervene in the browning of white adipose tissue. | Review article |
| Chris McGlory, PhD, Mark T. von Allmen, MSc, Tanner Stokes, BSc Robert W. Morton, PhD Amy J. et al. | Failed Recovery of Glycemic Control and Myofibrillar Protein Synthesis With 2 wk of Physical Inactivity in Overweight, Prediabetic Older Adults | 2018 | It was demonstrated that only 2 weeks of physical inactivity, could affect postprandial glycemia, generating the instauration of a diabetic phenotype. This decrease of insulin sensitivity is exacerbated with age. | Research article |
| Pedro L. Valenzuela, Pedro Carrera-Bastos, Beatriz G. Gálvez, Gema Ruiz-Hurtado, José M. Ordovas, Luis M. Ruilope, Alejandro Lucia | Lifestyle interventions for the prevention and treatment of hypertension | 2020 | Myokines offer direct and indirect benefits in cardiovascular phenotypes, including those related with blood pressure (BP). Especially irisin has shown that it decreases BP directly. | Review article |
| Rafael A. Casuso, Jesús R. Huertas | Mitochondrial Functionality in Inflammatory Pathology-Modulatory Role of Physical Activity | 2021 | Exercise induces adaptations at the mitochondrial level which help fight viral infections. Moderate intensity exercise increases immune cell level, an important data in facing the COVID-19 crisis and future pandemics. | Review article |
| Jimena Giudice, Joan M. Taylor | Muscle as a paracrine and endocrine organ | 2017 | Irisin decreases obesity through the process of browning of white adipose tissue, activates thermogenesis and promotes energy wasting.. | Review article |
| Claire Laurens, Audrey Bergouignan, Cedric Moro | Exercise-Released Myokines in the Control of Energy Metabolism | 2020 | We highlight the therapeutic potential of myokines and its importance in the interaction of muscle with other organs. The amount of muscle contractions is directly proportional with health benefits. | Review article |
| Carolina Archundia‑Herrera, Maciste Macias‑Cervantes, Bernardo Ruiz‑Muñoz, Katya Vargas‑Ortiz, Carlos Kornhauser, Victoriano Perez‑Vazquez | Muscle irisin response to aerobic vs HIIT in overweight female adolescents | 2017 | Irisin induces browning of white adipose tissue, resulting in an increase in thermogenesis. The expression of this protein in muscle is later incremented after a session of HIIT. | Research article Cross-sectional study |
| Steven S. Welc and Thomas L. Clanton | The regulation of interleukin-6 implicates skeletal muscle as an integrative stress sensor and endocrine organ | 2017 | IL-6 possesses functions of protection and regeneration in the human body, particularly in the acute stress scenarios. | Review article |
| Alejandro Abarca | Ejercicio como tratamiento anti-inflamatorio | 2016 | IL-6 increases the concentration of fatty acids during exercise through its lipolytic activity, furthermore it could help in the regulation of glucose homeostasis by altering the hepatic production of glycogen and/or the muscular capture of glucose.. | Literature review |
| Source: Own elaboration. |
DISCUSSION
MUSCLE AND PLASTICITY
The muscle is a tissue structurally organized and functional, acting as the main reservoir of amino
acids, hosting approximately 75% of body protein (3,8,10).
Muscle tissue is highly malleable, plastic (3,8), and
with a great capacity of adaptation and regeneration in the face of diverse stimuli such as nutrient
ingestion, restriction or increase of calories, physical activity, diseases, hormones, physical stress
and cytokines (11).
It should be emphasized that nutrition and exercise are key for increasing and preserving muscle
mass (3,8,12,13). Muscle
maintenance through life is necessary for health and wellbeing and recover from diseases (3). Furthermore, it guarantees physical independence, improves quality of life
(13) and prevents comorbidities, such as insulin resistance (12). Likewise, muscle strength has been seen as a predictor of mortality,
disability, and weakness in older adults (13,14).
The normal physiological aging behavior causes a decrease in muscle mass (13,14,15). During the aging process type II
muscle fiber atrophy occurs. Furthermore, there is a change from type II to type I fibers, meaning, from
fast to slow (3).
Under optimal conditions, the musculoskeletal system remains stable during the lifetime until
approximately 50 years, afterwards muscle loss descends progressively around 1% per year. After 70 years
of age, it increases to 1.3% - 2.4% and after 80 years of age it could reach 5% per year (13,14,15).
The aforementioned conceptualizes what is known as sarcopenia. The word sarcopenia is derived
from the Greek: sarx which means flesh, and penia: loss. It is defined as a chronic, degenerative
muscular disorder (8), generalized, progressive, associated with age, that is
characterized by decrease in musculoskeletal mass, muscle strength, and its functionality (13).
The mechanisms behind sarcopenia are multifactorial involving intrinsic factors such as
imbalance between protein synthesis and protein catabolism, hormone alterations, chronic inflammation,
neurodegenerative conditions, genetic factors, ectopic fat deposits and mitochondrial dysfunction
(12,15,16). As well as exogenous factors, for
example, nutritional deficiencies and physical inactivity. While it is true that sarcopenia is a process
parallel to age, a poor lifestyle could eventually accelerate this process (8,12,14).
A cross-sectional study carried out in Portugal with a total of 3493 older adults (1166 men,
2327 women) evidenced that a decrease in muscle mass and strength generates 1.65 to 2.2 greater risk of
becoming physically dependent (17,18)
On the other hand, a reduction in muscle mass and strength leads to effects in the locomotor
function of the musculoskeletal system, predisposing to alterations in glycemia and blood lipid levels,
particularly in state of obesity (19).
Scientific evidence exists where only 2 weeks of physical inactivity (650- 1000 steps per day),
as occurs in complete bedrest or hospitalizations, could cause important changes in the muscular level.
This leads to the introduction of insulin resistance and an increased risk of developing type 2 diabetes
mellitus, in young people as well as older adults (20).
Consequently, sarcopenia negatively impacts the economic level as well, since it increases
health care costs due to comorbidity worsening or complications, in addition to major risk of
hospitalizations (13), considered a public health problem (8).
THE MUSCLE AS AN ENDOCRINE ORGAN
The muscle is highly vascularized and possesses abilities to secrete myokines (22). During physical activity and exercise, repetitive contractions take
place which generate an adaptation to muscle cells, strengthening its contractile and metabolic
properties. This contractile activity is key for the production and release of myokines (11). These peptides not only participate in the regulation of energy demands,
but also benefit cardiovascular, metabolic and mental health (23).
Myokines regulate the muscular function and adaptation in oxidation, hypertrophy, and
angiogenesis (7). Furthermore, they are channels of communication between
muscle and different organs (23). By the way, two of the main positive
consequences of this molecular link between tissues are: a) decrease of systemic inflammation and with
this reduction of sarcopenia and accumulation of visceral fat, and b) greater sensitivity to insulin
(23).
There have been over 650 myokines identified (5). Below, we will
mention some of the more relevant and their implications in chronic diseases.
Interleukin-6 (IL-6):
Originally this interleukin was considered as a proinflammatory cytokine, however, in recent years,
studies have demonstrated its anti-inflammatory properties (11,24). Muscular contraction induced by physical activity generates and increase
in genetic expression and release of IL-6 by the myocyte (6,23), contributing with the homeostasis of glucose (8). The plasmatic levels of IL-6 can increase up to 100 times, depending on
the volume, intensity and density of physical exercise (6,23).
Some of the target organs for circulating IL-6 are the liver, pancreas and adipose tissue. IL-6
optimizes the viability of pancreatic β cells, improves insulin sensitivity and favors lipolysis in
adipose tissue (7,22). Furthermore, it has been seen to
stimulate hepatic gluconeogenesis, glucogenesis and glucose release (7,11).
On the other hand, this myokine, through alternative pathways, promotes the activation of
macrophages, which are involved in the protection against inflammation and insulin resistance secondary
to obesity (7).
Immediately after performing physical exercise, released IL-6 significantly increases the levels
of IL-10 and the interleukin 1 receptor antagonist receptor (IL-1Ra). These changes are characterized by
having an anti-inflammatory effect (6,11).
A very interesting data is that this anti-inflammatory effect is more evident in certain
pathological conditions, especially in those which inflammatory cytokines and CRP are found 2 to 3 times
above the basal levels, such as atherosclerosis, type II diabetes, obesity and cardiac insufficiency
(6).
Overall, IL-6 improves glycemic control, favors fat loss, suppresses tumoral growth and
collaborates in the maintenance of muscle mass (7).
Interleukin 15 (IL-15):
In humans, this interleukin is able to be expressed in various cells and tissues, including the
musculoskeletal system, epithelial cells, monocytes, and dendritic cells (9).
It is released at the muscular level mainly after strength and resistance physical exercise (2,9).
IL-15 has been characterized by its anabolic properties in the skeletal muscle (6,11), its ability to induce hypertrophy has been demonstrated
though the reduction of protein degradation in sarcopenia and cachexia, emphasizing this therapeutic
power (6,11). Another interesting data that reaffirms this
concept is that the plasma levels of IL-15 decrease with age, studies report that older adults with
sarcopenia have significantly lower levels of this myokine (9).
Parallel to this, IL-15 has been found to inhibit tumor necrosis factor α (TNF-α) in the muscle
during the states of cachexia. In addition, a decrease of this cytokine has been associated with
autoimmune disease such as rheumatoid arthritis and leukemia (6).
Myostatin:
It was the first identified myokine, despite not being called a myokine during its discovery era
(7). The main function of myostatin is to negatively regulate muscle mass
(7,11). It is said that this cytokine is “inverse” compared to
other muscular cytokines, in aerobic as well as strength and/or resistance exercise, decreases its
expression in myocytes considerably (9,11). Its ablation
generates muscular hypertrophy, increases muscle strength and improves bone architecture (24).
Patients with cardiac insufficiency and cancer possess high levels of this protein, which could
eventually result in cachexia. However, during physical activity these levels decrease, which
illustrates the anti-catabolic benefit in these types of patients (6,9). The same result is observed in patients with chronic renal disease
(9).
Myostatin has received new attention due to its potential for treatment of metabolic syndrome,
since the levels of this substance are found elevated in patients with type 2 diabetes and women with
obesity (7).
Muscle – adipose tissue and irisin:
A tissue that maintains a tight “communication” with the muscle is adipose tissue. Adipose tissue is
constituted by 2 types of fat: white fat and brown fat. White stores the excess energy and secretes
innumerable proinflammatory substances, meaning, in general terms it could be said that it is
metabolically unfavorable. However, brown fat is beneficial (22), since it
has the capacity of regulating thermogenesis due to its high concentration of mitochondria (6), which means it consumes accumulated energy and therefore the levels of
triglycerides decrease (22).
The muscle – adipose tissue communication during muscle contractions provoke decrease in
adiposity, increase in thermogenesis due to the increments in adrenergic activity, browning of white fat
tissue, and increase in lipolysis (6,22). This browning
process occurs mainly by the myokine called irisin, which joins a receptor in the adipocytes, favoring
the phenotypic modification of fat and with that the increase in energy waste. This thermogenic effect
is
mediated by the expression of an uncoupling protein present in mitochondria, also known as
thermogenin or UCP1 (uncoupling protein 1) (6) Figure 1
summarizes this process clearly.
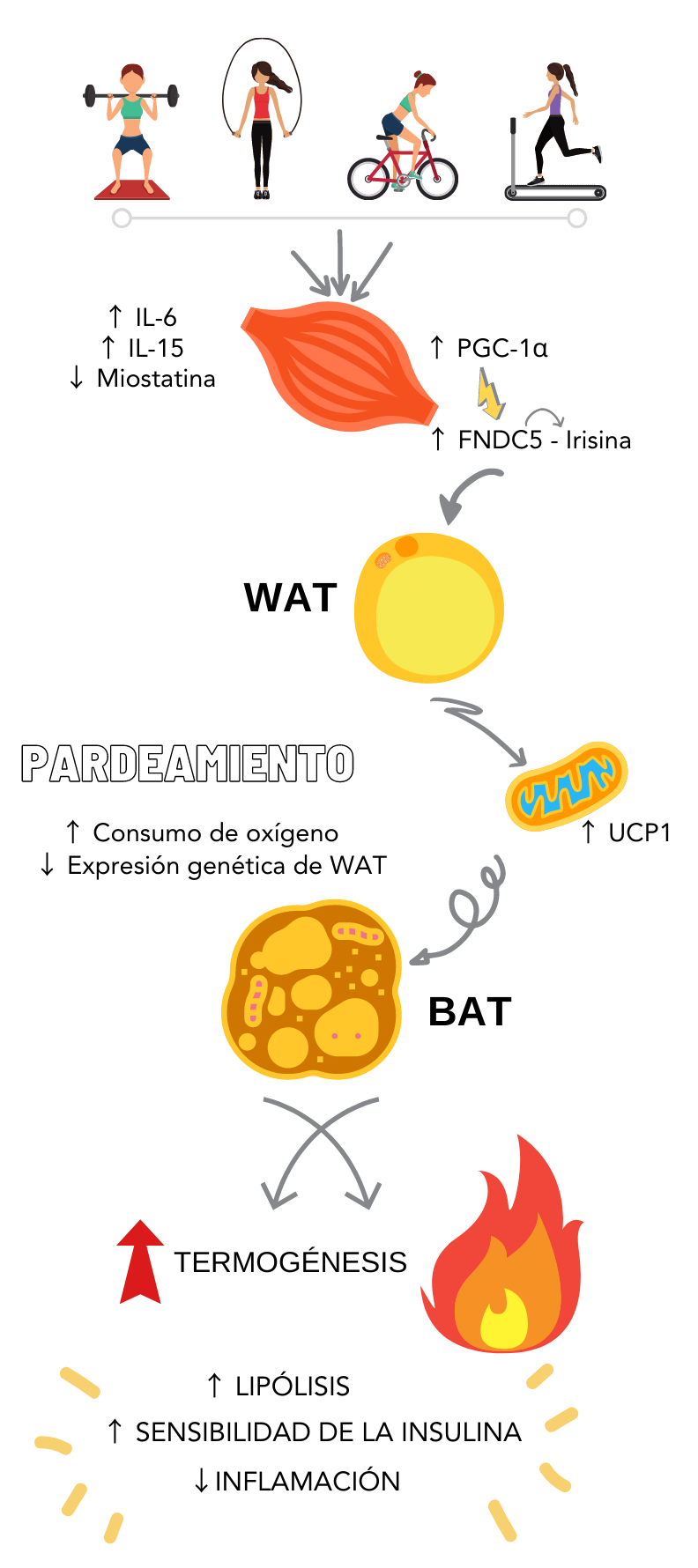
Additionally, it has been reported that it protects pancreatic function through the stimulation of
pancreatic β cell proliferation and protection in face of apoptosis (24).
Irisin production is greater in cardiac muscle than in skeletal muscle. However, aerobic exercise favors
its genetic expression generating an increase in plasma level (9).
On the other hand, several studies have been documented trying to demonstrate if irisin levels
increase more significantly with aerobic training versus anaerobic. However, there are discrepancies
between this evidence, which explains through the technique used to measure serum levels of irisin.
Still, there is overwhelming scientific evidence that both types of exercise could increase circulating
irisin (25).
Muscle-brain:
Physical activity represents one of the most effective strategies to reduce prevalence and incidence of
depression, cognitive impairment, neurodegenerative disorders (1). Parallel to
this it has a positive impact on stress and anxiety (5).
Exercise influences the hippocampus more than any other anatomical brain structure (5) through numerous myokines including cathepsin B, irisin, brain derived
neurotrophic factor (BDNF), fibroblastic growth 21 (FGF-21), which play an important role in the
muscle-brain interaction (22,26). The same, could measure
neurological functions such as: cognitive development, memory, neuroplasticity, motor coordination,
sleep, mood (26), learning ability and memory (24). At the same time, offer neuroprotection and decrease in brain
inflammation (26).
One of the noted benefits of physical exercise is central and peripheral induction of BDNF
(26), which plays a dominant role in this muscle-brain interaction (5). BDNF is a growth factor that belongs to the neurotrophin family, which
look out for the neuronal survival and synaptic plasticity (11,26).
It is important to mention that patients carrier of Alzheimer’s disease, with depression,
obesity, and type 2 diabetes, possess reduced serum levels of BDNF (11).
The type 1 insulin growth factor (IGF-1), mainly produced by the liver, is able of crossing the
hematoencephalic barrier and acts as mediator of attributable changes in physical activity in
neurogenesis of the hippocampus and expression of BDNF. The formation and maintenance of dendritic
spines at the hippocampus neuronal level in basal conditions, depend on BDNF induced by PGC-1α
(coactivator of 1α of the gamma activated receptor of the peroxisome proliferator) after exercise. In
the neuronal tissue, PGC-1α is a potent suppressor of the reactive oxygen species (ROS), which explains
its neuroprotective effect (26).
Muscle-cancer:
Epidemiological studies suggest that physical activity carried out in times of leisure reduce the risk
of 13 different types of cancer (5). People that are physically active after
their diagnosis of prostate, colon, and breast cancer have a greater survival rate than those that are
sedentary with the same type of neoplasia (5).
Exercise stimulates NK cells (natural killer), which play a central role in reducing tumoral
growth. Furthermore, IL-6 and IL-5 regulate the maturity and redistribution of NK cells (7).
PHYSICAL EXERCISE: PREVENTION AND TREATMENT
Given the anti-inflammatory effects of myokines as a response to physical exercise, it is clear that
physical inactivity leads to inflammation and increase in adipose tissue deposits (26).
In fact, the association between physical inactivity and visceral fat has been clearly
established, which is more inflammatory than subcutaneous, leads to systemic chronic inflammation that
predisposes to atherosclerosis, dyslipidemia, insulin resistance, neurodegeneration, anemia, and muscle
degeneration. These pathologies are factors that provoke inactivity. The lack of movement generates more
inflammation, and this continues to open the path to more chronic diseases, establishing a chronic
vicious cycle (5).
Given the above, it is clearer that physical exercise helps improve the body composition in
individuals with overweight and obesity (6). Furthermore, it decreases
visceral fat, which includes pericardial fat (5). It is evident that the
therapeutic strategy for sarcopenia is still physical exercise. And with these additional benefits are
obtained such as increasing strength, increasing basal metabolic rate, decreasing blood pressure, and
optimizing blood lipid levels (12).
It’s important to note that during decades the importance of aerobic exercise has been
mentioned. However, recently it has been demonstrated that physical activity that involves strength and
resistance could be just as effective for reducing morbidity and optimizing health (18).
Given the above, different exercise modalities have been demonstrated for blood pressure
decrease. Studies support that all type of training, whether it is aerobic, or anaerobic (resistance or
strength), or a combination of both, are equally effective for reducing pressure in persons with
arterial hypertension (27).
Delving into this more than what was described in the prior paragraph, and despite that
cardiovascular exercise is the most prescribed for this type of patients, the benefits of strength
training exercise such as weight lifting, or a combination of aerobic exercise and resistance exercise,
are similar and even superior in the decrease of arterial pressure, The following reductions have been
registered: 8.7 mmHg in cardiovascular exercises, 7.2 mmHg in resistance and 13.5 mmHg in the
combination of both (27).
There is evidence supporting that regular exercise, even without important weight loss, the
levels of leptin decrease. Similarly, intense acute exercise reduces the circulating levels of
proinflammatory adipokines tightly linked with insulin resistance and obesity. For this, physical
exercise offers benefits to individuals with arterial hypertension, as well as other chronic diseases,
secondary to positive effects generated by the increase of myokines and reduction of adipokines during
and after training (27).
On the other hand, the systemic response to myokines is greater after physical exercise with a
greater level of fiber breakdown, for example in resistance training, taken in downhill slopes and
eccentric exercises. In the same manner, we have observed an important increase in levels of myokines
after training of long duration and vigorous intensity and much less pronounced in physical activity of
moderate intensity (7).
Physical activity impacts glucose and lipid homeostasis, altering the composition of muscle
fibers. Specifically, strength and resistance training favor the growth of muscle tissue, and at the
same time increase oxidative and glycolytic capacity of fast fibers. It is scientifically documented
that diabetics with a resistance exercise regimen have improved their insulin sensitivity. Likewise, in
obese patients when gaining muscle mass, increase their basal metabolic rate (28).
Lastly, the best road to maintain a healthy musculoskeletal system is increasing and maintaining
a good muscle mass through physical exercise, and with that not just guarantee longevity but also
quality of life (9). Furthermore, science continues to reaffirm that physical
exercise is effective not just as prevention but also as medical treatment for different chronic
diseases (23).
CONCLUSION
Muscular activity is crucial for health and physical-mental wellbeing. Recently, with the discovery of myokines, the muscle has been involved in the maintenance of metabolic and cognitive homeostasis. If the muscle’s endocrine function is not sufficiently stimulated with physical exercise, the production of myokines necessary for the optimal functioning of organs would not be adequate, generating as a consequence the development of diseases. The knowledge on cytokines released by the muscle in response to physical exercise, is of key help for the prescription of exercise in an individualized manner and at the measure of each patient for the treatment of diseases associated with inactivity.
Authorship contributions: The authors participated with the idea genesis, project design,
data collection and interpretation, results analysis and manuscript preparation of this resarch
work.
Funding sources: Self-financed.
Conflicts of Interest: The authors declare not having conflicts of interest.
Received: August 16, 2021
Approved: November 22, 2021
Correspondence: Amelia Vargas Pacheco
Address: San José, Costa Rica. Hospital La California.
Telephone number: +506 88826960
E-mail: doctora.ame@gmail.com
REFERENCES
