CASO CLÍNICO
REVISTA DE LA FACULTAD DE MEDICINA HUMANA 2022 - Universidad Ricardo Palma10.25176/RFMH.v22i4.4762
GASTRIC ADENOMYOMA IN ADULT A CASE REPORT
ADENOMIOMA GÁSTRICO EN ADULTO: REPORTE DE CASO
Zamora Gonzales, Pedro Luis1,2,3,a, Somocurcio Peralta, José1,b, Yagui Uku, Carmen Elena3,c, Goicochea Arévalo, Ronald1,d
1Hospital Nacional Edgardo Rebagliati Martins. Lima – Perú.
2Facultad de Medicina, Universidad Nacional Mayor de San Marcos.
3Hospital Nacional Daniel A. Carrión, Callao, Perú.
aMaster's degree - General Surgeon
bMaster's degree - clinical pathologist
cMaster's degree - clinical pathologist
dGeneral Surgeon
ABSTRACT
Objective: Report the clinical case of Gastric Adenomyoma as etiology of pyloric syndrome. Clinical case: We present a 52 years old man who attended the emergency room due pyloric syndrome and who underwent a distal gastrectomy. The pathological report revealed a gastric adenomyoma with ulcerative lesion. Discussion: This pathology is an unusual finding, and the literature describe it with extremely low rate of malignant transformation. Conclusion: Gastric adenomyoma is a infrequent pathology and should be taken into account when endoscopic studies are not enough to determine the etiology of a pyloric syndrome.
Keywords: Adenomyoma; Stomach; Pylorus. (Source: MeSH NLM).
RESUMEN
Objetivo: Reportar el caso clínico de Adenomioma gástrico como etiología de un síndrome pilórico. Caso Clínico: Presentamos el caso de un varón de 52 años que acudió a emergencia por un síndrome pilórico quien fue sometido a una gastrectomía distal. El reporte de anatomía patológica revelo un Adenomioma gástrico con lesión ulcerativa múltiple. Discusión: Esta patología es un hallazgo inusual, siendo extremadamente baja su tasa de transformación maligna. Conclusiones: El Adenomioma gástrico es una patología infrecuente que debe ser tenida en cuenta cuando los estudios endoscópicos no logran definir la etiología de un síndrome pilórico.
Palabras Clave: Adenomioma; Estomago; Píloro. (fuente: DeCS BIREME).
INTRODUCTION
Gastric adenomyoma is a rare benign pathology characterized by ducts and glands surrounded by smooth muscle stroma(1-5). The ages of presentation are variable, presenting from neonates to the elderly(2). 52 cases have been described in the literature up to 2017(1,4). The most frequent age of presentation is between the fourth and sixth decades of life (1,4) without gender predilection(1) and may be associated with annular pancreas, Gardner syndrome with duodenal adenomas and gastric duplication(1). The clinical presentation is variable, and may include nausea, vomiting, epigastric pain, even hematemesis, anemia and melena(1,3,4). Despite being considered a benign disease, cases of malignant transformation have been described in up to 2%(4). The importance of taking this pathology into account lies above all when the endoscopic findings are inconclusive in the presence of pyloric syndrome that does not show malignant disease or other types of obvious obstructive lesions.
DESCRIPTION OF THE CLINICAL CASE
A 52-year-old male, natural from Iquitos, with no relevant history, goes to the emergency room reporting 3 days of epigastric pain, nausea, vomiting and the inability to eliminate flatus for 2 days; also loses weight of 7kg in 1 month and oral intolerance to solids and liquids; also, increased appetite. Examination shows marked distension of the upper abdomen with tympanism, without peritoneal reaction; A simple abdominal X-ray was performed, showing great dilatation of the gastric chamber with no signs of lower intestinal obstruction or air-fluid levels. It is concluded that the patient presents emesis due to a pyloric syndrome to rule out an infiltrative lesion; hospitalization is indicated to complete studies.
The total abdominal tomography with intravenous contrast evidenced: stomach with distention and food content inside, impresses conditioned by an obstructive process located at the pyloric level that is thickened up to 16mm with diffuse mucosal enhancement of probable inflammatory cause, the adjacent fat shows diffuse striation ; concluding mural thickening at the level of the pyloric region with diffuse mucosal enhancement conditioning gastric obstructive process, consider high suspicion of inflammatory process, the possibility of neoplastic infiltrative process is not discard.
An upper gastrointestinal endoscopy was performed where a linear ulcer with regular edges and a fibrinous bed was described at the level of the gastric angle, which were biopsied, as well as deformed antrum due to healing of the lesser curvature ulcer, pylorus occluded due to healing of the lesser curvature ulcer. Duodenal bulb and second duodenal portion not observed; gastric ulcer was concluded that deforms and occludes pylorus without signs of active bleeding. The pathological anatomy described severe active plasmacytic chronic gastritis with edema and fibrosis of the lamina propria, regenerative epithelium with mild dysplasia, necrotic ulcer fundus with shadow cells.
Preoperative studies describe: Leukocytes 8.03x103/uL, Hemoglobin 12.1g/dL, Platelets 320x103/uL, blood group O positive, Albumin 4.1g/dL, Creatinine 0.69mg/dL, Glucose 94mg/dL, Urea 41.2mg/dL.
After the endoscopic studies and the pathological anatomy reports do not conclude the etiology of the pyloric syndrome, it is decided to perform a Radical Distal Gastrectomy with D2 Lymphadenectomy with reconstruction by Y Roux Gastrojejunostomy due to a pre-pyloric gastric inflammatory process that conditions partial obstruction of the pylorus, where the possibility of a neoplastic infiltrative process is not ruled out. The findings showed a pre-pyloric gastric tumor with lesser curvature of 4x3cm in diameter, hard that occludes and compresses the pylorus, with serosal retraction of the posterior gastric wall that conditions gastric chamber dilatation. No liver or peritoneal lesions or free fluid. After the surgical procedure, the patient evolved favorably and was discharged on the eleventh postoperative day with good oral tolerance.
The pathological anatomy report of the operative piece described: Gastric adenomyoma with multiple ulcerative lesion, fibrosis and serosal retraction with hypertrophic arterial vessels that reach the submucosa (Figure 1) Peri-ulcerative regenerative epithelium and granulation tissue with isolated giant cells of the foreign body type . Chronic follicular gastritis with follicular mantle hyperplasia, free surgical margins – free omentum, free perigastric lymph nodes 0/26 with adenitis and lymphatic congestion.
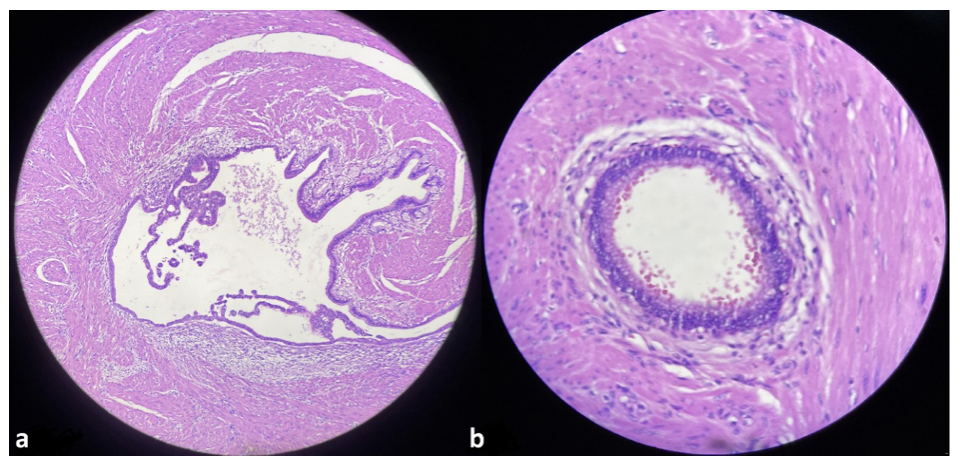
Groups of glands surrounded by bundles of smooth muscle. a(x10) b(x40)
DISCUSSION
Gastric adenomyoma is a rare benign lesion composed of ducts and glands surrounded by smooth muscle stroma(1-5). Also known as myoglandular hamartoma, adenomyomatous hamartoma or myoepithelial hamartoma(2,3,5). It is usually found in the antrum and pylorus and, when the adenomyoma is extragastric, it is located in the duodenum and jejunum. For several authors, the location of adenomyomas is gastric (25-38%), duodenum (17-36%) and jejunum (15-21%), and the location in the stomach is: antrum (85%), pylorus (15% )(1,3) and exceptionally in the body(1).
Magnus Alsleben was the first to describe this entity in 1903, the product of 5 cases found incidentally in necropsies(5). Various authors consider this entity to be a hamartoma while others consider it a variant of a heterotopic pancreas(1,2). In some cases it coexists with heterotopic pancreas and in other cases it shows communication between the gastric lumen and the epithelial component of the lesion(1).
In 1909, Heinrich described 3 histological subtypes of heterotopic pancreas, considering the third type as “adenomyoma”, while Matsushita described his echoendoscopic findings and related them to Heinrich's description as follows(1):
Type 1: Exocrine and Endocrine Pancreas (Heterotopic Pancreas); Echo endoscopy shows indistinct margins, heterogeneous appearance and location between the third and fourth layers.
Type 2: Exocrine Pancreas without islets (Heterotopic Pancreas); echo endoscopy shows similar to type 1 but localization only in the third layer.
Type 3: Only ducts (adenomyoma); Echo endoscopy shows a small anechoic nucleus with thickening of the fourth layer.
The ages of presentation are variable, with the youngest patient being a 1-week-old neonate and the oldest being an 81-year-old person(2); To date, 52 cases have been described in the literature up to 2017(1,4) with the most frequent age of presentation being between the fourth and sixth decade of life(1,4) with no gender predilection(1). Associated conditions may include annular pancreas, Gardner syndrome with duodenal adenomas, and gastric duplication(1).
The clinic is diverse, and can range from the asymptomatic which are discovered during a laparoscopy or necropsies(1,3), with the most reported cases being those associated with non-specific symptoms such as nausea, vomiting, epigastric pain, hematemesis, anemia, melena(1,3,4) and even reaching intermittent pyloric obstruction(3). Likewise, cases of secondary peritonitis due to perforation have been described. Making a preoperative diagnosis is difficult, because most of the symptoms are not specific, in addition to the few cases described in the literature(1,3).
For diagnosis, endoscopy is not usually very effective because it manifests as a submucosal lesion, in addition to not being able to differentiate between hamartoma and leiomyoma, melanoma, lymphoma, carcinoid tumor, Kaposi's sarcoma, GIST or eosinophilic granuloma(3). For which, all these injuries must end up in surgical management to ensure a correct diagnosis(2-4).
Despite being considered a benign lesion, the potential for malignant transformation does exist(1-3) being less than 2%(4). The literature has described a case of gastric adenomyosis with malignant transformation of its glandular center(2). The use of endoscopic technologies for the resection of this type of lesion has not been reported; however, in retrospective studies it has been determined that these lesions have been resected using this technique without further complications(2,4). Most of the reviewed series indicate the open or laparoscopic surgical approach as the ideal management option.
CONCLUSIONS
Gastric adenomyoma is a rare benign pathology, difficult to diagnose clinically, which is usually diagnosed after the study of surgical specimens. It should be considered as a diagnostic presumption when previous studies are inconclusive; likewise, surgical management should not be delayed due to a lack of etiology, since this can generate unnecessary delays that could lead to clinical deterioration in patients.
Authorship contributions: The authors participated in the genesis of the idea, project design, data collection and interpretation, and preparation of the manuscript of this research work.
Funding sources: Self-financed .
Conflicts of interest: There was no conflicts of interest.
Received: February 09, 2022
Approved: July 27, 2022
Correspondence: Goicochea Arévalo, Ronald.
Address: Jirón Joaquín Bernal 595 Dpto. 701. Lince – Lima - Perú.
Telephone number: 945666530
E-mail: ronald_goicochea@hotmail.com
Article published by the Journal of the faculty of Human Medicine of the Ricardo Palma University. It is an open access article, distributed under the terms of the Creatvie Commons license: Creative Commons Attribution 4.0 International, CC BY 4.0(https://creativecommons.org/licenses/by/1.0/), that allows non-commercial use, distribution and reproduction in any medium, provided that the original work is duly cited. For commercial use, please contact revista.medicina@urp.edu.pe.
REFERENCES
