Evaluation of three brands of rapid tests against blood samples for the detection of antibodies against HIV
Evaluación de tres marcas de pruebas rápidas frente a muestras de sangre para la detección de anticuerpos contra VIH
Abstract
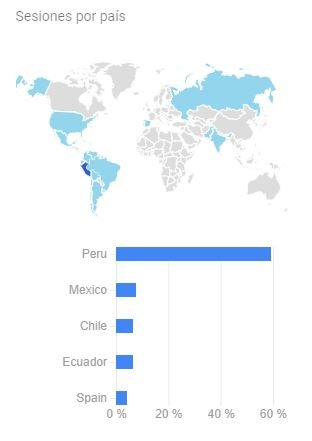
One of the important factors to reduce the spread of the human immunodeficiency virus (HIV) in Peru is the accessibility of diagnosis through rapid tests (RP), which are applied even in the most remote communities. However, during the five years 2011 - 2015, health establishments acquired and used more than 10 different brands, generating confusion in applying the methodology described in the inserts and consequently erroneous results (false positives and false negatives).
Downloads
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2021 Revista de la Facultad de Medicina Humana

This work is licensed under a Creative Commons Attribution 4.0 International License.